Digital centerpiece in trials
The high performance technology platform for multi-channel data sourcing in medical science.
Your central access to clinical data
Clinical research undergoes rapid changes, driven by digitalization, big data analytics and machine learning. Yet we stay focused on the baseline: valid data - in digital formats. Created in extensive scopes and bandwidths, in many formats, collected from a huge variety of sources.
INTEGRATIONS
One Platform for your Study
Integrating multiple data sources and functionalities into clinical studies is the purpose of Alcedis Platforms™. What can be imagined is build into one piece. The REST-API interface of Alcedis Platforms™ is the key to integrate and align our own and third-party solutions into the Alcedis ecosystem.
Get integrated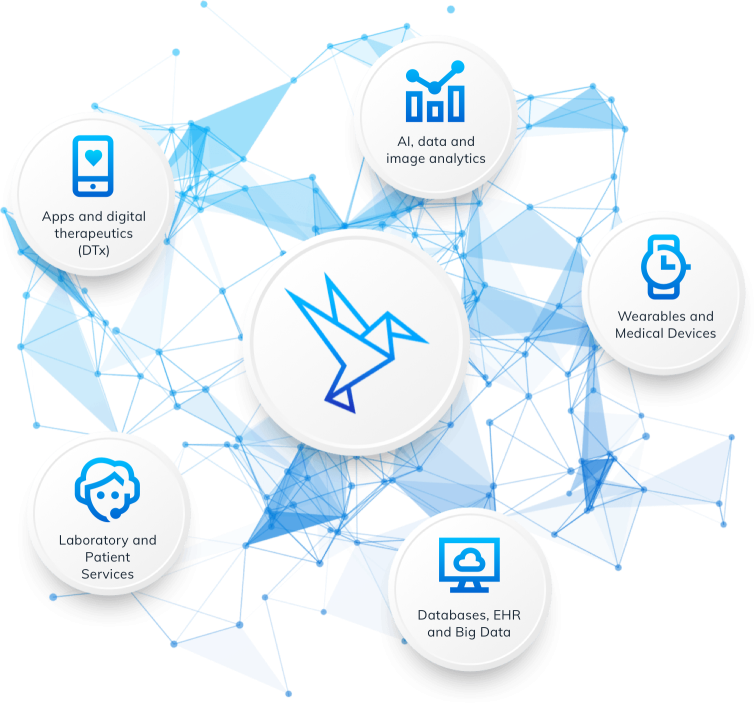
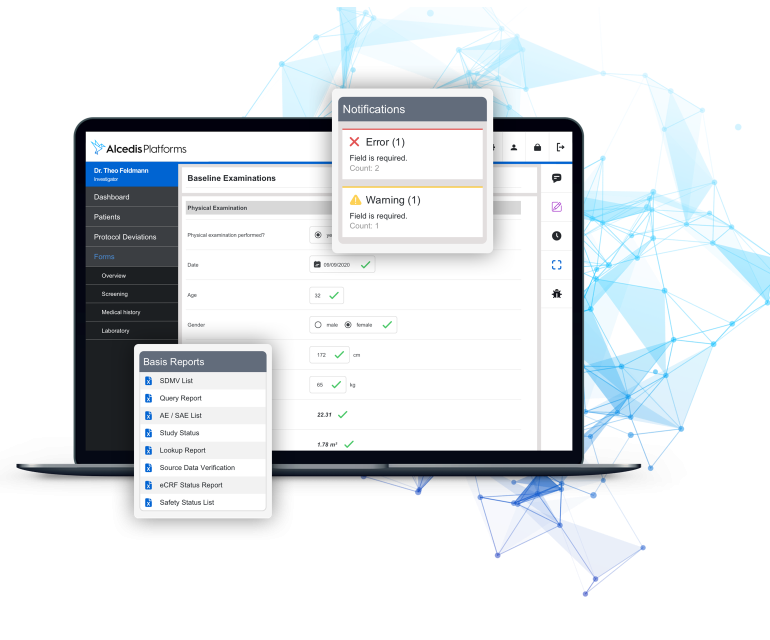
eCRF
Flexible, intuitive and secure
- Starting with a complex protocol or a simple vision, we are the experts to deliver a custom-built eCRF within short set-up times
- Creating powerful and disruptive research environments since we launched Germany's first eCRF in 1998
- Innovative user interfaces and databases safely hosted via our in-house cloud computing center in Germany
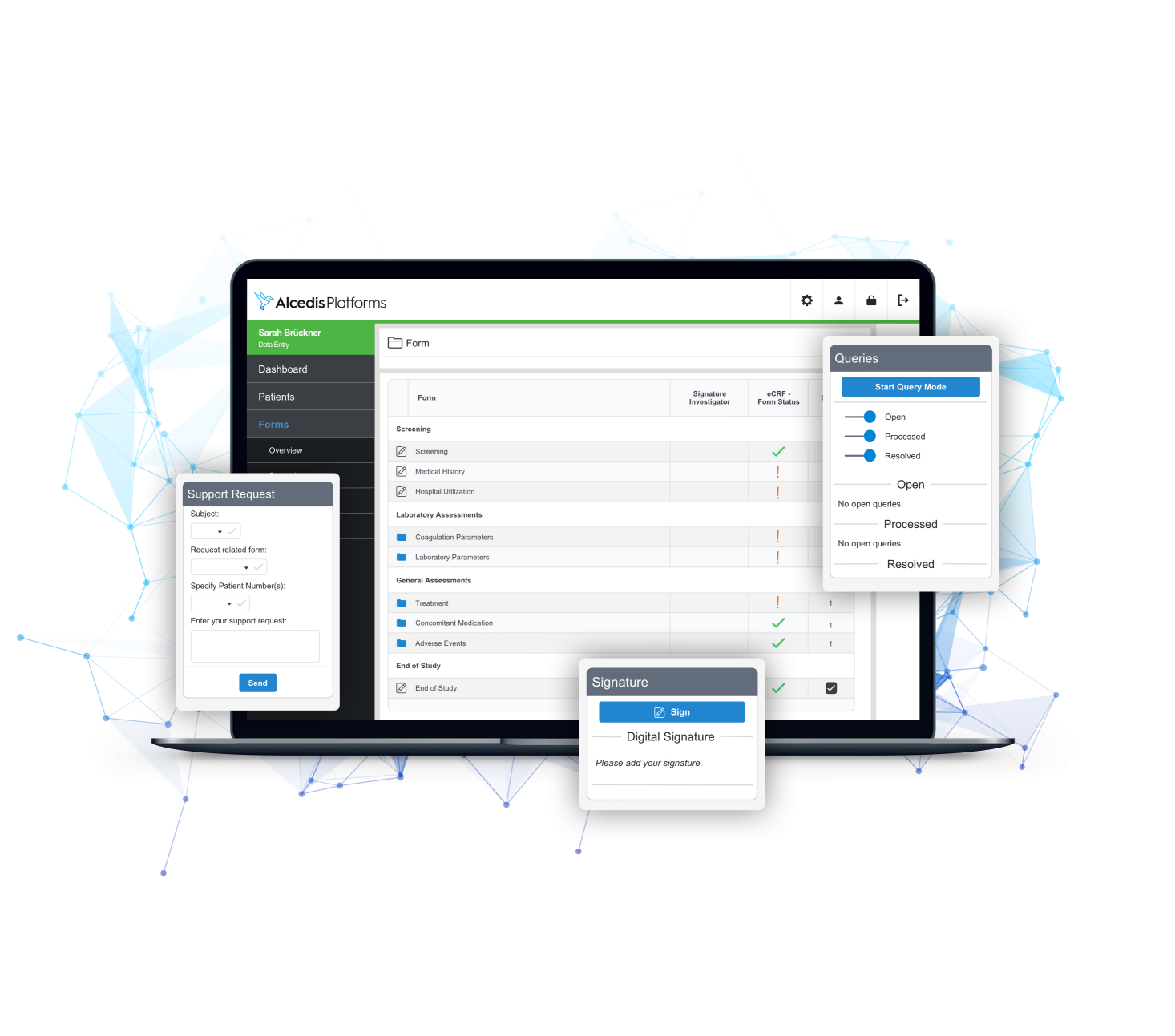
PLUGINS
Best of Class for Maximum Synergy
- Robust off-the-shelf extensions for the most common and important processes
- Reduced set-up timelines and maximum cost efficiency
- Benefit from our experience in digitalizing workflows
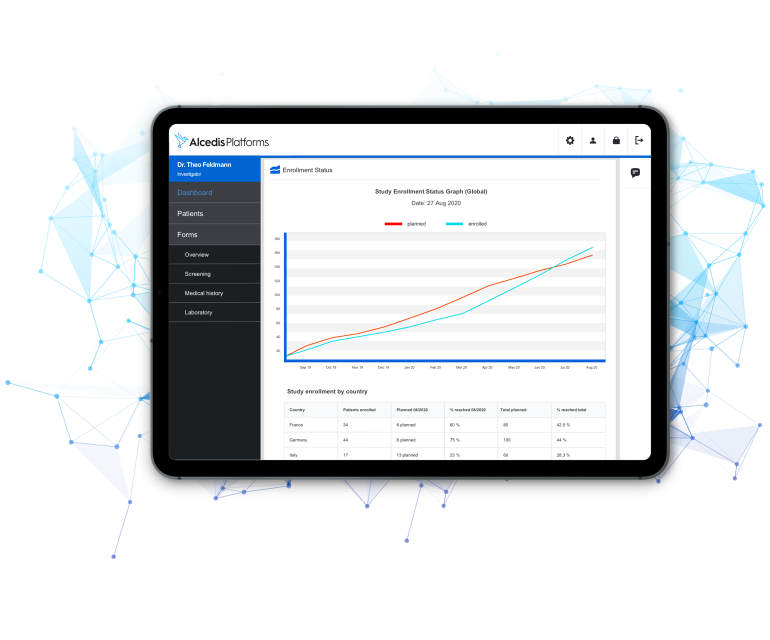
MOBILE
Extend your Platform to any Mobile Device
- Simple recording of study data for participants in clinical studies (e.g. Quality of Life Data)
- Completion of important tasks such as editing forms or calling up reports on mobile devices
- Current information on relevant performance indicators and study progress for project managers
Launch your Alcedis Platform™!
Let us create a high-performance environment for science that matters. Together!
Contact us




